2010
Wiederhold K., Kloepper T.H., Walter A.M., Stein A., Kienle N., Sorensen J.B., and
Fasshauer D
A coiled-coil trigger site is essential for rapid binding of synaptobrevin to the SNARE
acceptor complex.
J Biol Chem. 285:21549-59
DOI / PMID
Walter A.M., Wiederhold K., Bruns D., Fasshauer D., and Sørensen J.B.
Synaptobrevin N-terminally bound to syntaxin-SNAP-25 defines the primed vesicle state in
regulated exocytosis.
Cell Biol. 188(3):401-13
DOI / PMID
2009
Winter U., Chen X., Fasshauer D
A conserved membrane attachment site in α-SNAP facilitates NSF-driven SNARE complex
disassembly.
J Biol Chem. 284(46):31817-26
DOI / PMID
Domanska M.K., Kiessling V., Stein A., Fasshauer D., Tamm L.K.
Single vesicle millisecond fusion kinetics reveals number of SNARE complexes optimal for
fast SNARE-mediated membrane fusion.
J Biol Chem. 284(46):32158-66
DOI / PMID
Radhakrishnan A., Stein A., Jahn R., Fasshauer D
The Ca2+ affinity of synapto-tagmin 1 is markedly increased by a specific
interaction of its C2B domain with phosphatidylinositol 4,5-bisphosphate.
J Biol Chem. 284:25749-60
DOI / PMID
Kienle N., Kloepper T.H., and Fasshauer D
Differences in the SNARE evolution of fungi and metazoa.
Biochem Soc Trans 37:787-91
DOI / PMID
Wiederhold K., Fasshauer D
Is assembly of the SNARE complex enough to fuel membrane fusion?
J Biol Chem. 284:13143-52
DOI / PMID / Comment in DOI / PMID
Kienle N., Kloepper T.H., and Fasshauer D
Phylogeny of the SNARE vesicle fusion machinery yields insights into the conservation of
the secretory pathway in fungi.
BMC Evolutionary Biology 9:19
DOI / PMID / SNARE Database
2008
Bar-On, D., Winter,U., Nachliel, E., Gutman, M., Fasshauer, D., Lang, T., and Ashery, U.
Imaging the assembly and disassembly kinetics of cis-SNARE complexes on native plasma
membranes.
FEBS Letters, 582:3563-8
DOI / PMID
Kloepper T.H., Kienle C.N., and Fasshauer D
SNAREing the basis of multicellularity: Consequences of protein family expansion during
evolution.
Mol.Biol.Evol. 25:2055-68
DOI / PMID / SNARE Database
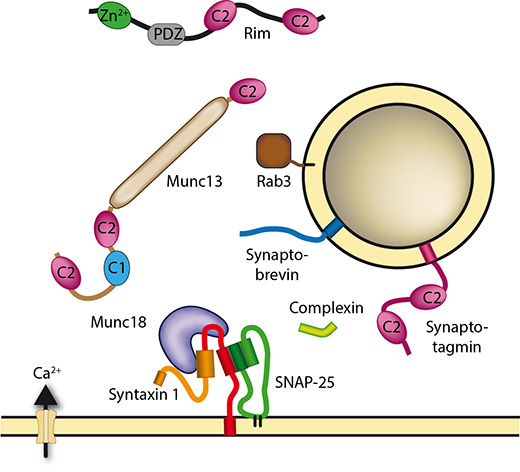
Burkhardt P., Hattendorf D. A., Weis W. I., Fasshauer D
Munc18a controls SNARE assembly through its interaction with the syntaxin N-peptide.
EMBO J. 27: 923-33
DOI / PMID
Barszczewski M., Chua J., Stein A., Winter U., Heintzmann R., Zilly F.E., Fasshauer D.,
Lang T., and Jahn R.
Binding of α-SNAP to syntaxin 1 blocks SNARE-dependent exocytosis.
Mol. Biol. Cell 19:776-84
DOI / PMID
2007
Stein, A., Radhakrishnan, A., Riedel, D., Fasshauer, D., and Jahn, R
Synaptotagmin activates membrane fusion through a Ca2+-dependent trans
interaction with phospholipids.
Nat. Struct. Mol. Biol. 14: 904-11
DOI / PMID
Fasshauer, D. & Jahn, R.
Budding insights on cell polarity (News & Views).
Nature Structural & Molecular Biology 14: 360-2
DOI / PMID
Kloepper, T.H., Kienle, C.N., and Fasshauer, D
An elaborate classification of SNARE proteins sheds light on the conservation of the
eukaryotic endomembrane system.
Mol. Biol. Cell 18: 3463-71
DOI / PMID / InCytes / SNARE Database
Siddiqui, T.J., Vites, O., Stein, A., Heintzmann, R. Jahn, R., and Fasshauer, D
Determinants of Synaptobrevin regulation in Membranes.
Mol. Biol. Cell 18: 2037-46
DOI / PMID / InCytes
Zwilling, D., Cypionka, A., Pohl, W., Fasshauer, D., Walla, P.J., Wahl, M.C., and Jahn R.
Early endosomal SNAREs form a structurally conserved SNARE complex and fuse liposomes
with multiple topologies.
EMBO J. 26: 9-18
DOI / PMID
2006
Pobbati, A., Stein, A., and Fasshauer, D
N- to C-terminal SNARE complex assembly promotes rapid membrane fusion.
Science 313: 673-6
DOI / PMID
Holt, M., Varoqueaux, F., Wiederhold, K., Takamori, S., Urlaub, H., and Fasshauer, D. and
Jahn, R.
Identification of SNAP-47, a novel Qbc-SNARE with ubiquitous expression.
J. Biol. Chem. 281: 17076-83
DOI / PMID
Sorensen JB., Wiederhold, K., Müller EM., Milosevic, I., Nagy, G., de Groot, BL.,
Grubmüller, H., and Fasshauer, D
Sequential N- to C-terminal ‘zipping-up’ of the SNARE complex drives priming and fusion
of secretory vesicles.
EMBO J. 25:955-66
DOI / PMID
2005
Nagy G., Milosevic I., Fasshauer D., Müller EM., de Groot BL., Lang T., Wilson MC.,
Sørensen JB.
Alternative Splicing of SNAP-25 Regulates Secretion through Nonconservative Substitutions
in the SNARE Domain.
Mol. Biol. Cell 12: 5675-85
DOI / PMID
2004
Pobbati A., Razeto A., Böddener M., Becker S., Fasshauer D
A structural basis for the inhibitory role of tomosyn in exocytosis.
J. Biol. Chem. 279: 47192-200 “JBC Paper of the week”
DOI / PMID
Fasshauer, D. and Margittai, M.
A transient N-terminal interaction of SNAP-25 and syntaxin nucleates SNARE assembly.
J. Biol. Chem. 279: 7613-21
DOI / PMID
2003
Margittai, M., Widengren, J., Schweinberger, E., Schröder, G.F., Felekyan, E., Haustein,
E., König, M., Fasshauer, D., Grubmüller, H., Jahn, R., Seidel, C.A.M.
Single-molecule fluorescence resonance energy transfer reveals a dynamic equilibrium
between closed and open conformations of syntaxin-1.
PNAS 100: 15516-21
DOI / PMID
Fasshauer D
Structural insights into the SNARE mechanism.
BBA - Molecular Cell Research. Special Issue: Membrane Fusion, 1641: 87-97
DOI / PMID
Hatsuzawa. K., Lang, T., Fasshauer, D., Bruns, D., and Jahn, R.
The R-SNARE motif of tomosyn forms SNARE core complexes with syntaxin 1 and SNAP-25 and
down-regulates exocytosis.
J. Biol. Chem. 278: 31159-66
DOI / PMID
Reidt, U., Wahl, M.C., Fasshauer, D., Horowitz, D.S., Lührmann, R., and Ficner, R.
Crystal structure of a complex between human spliceosomal cyclophilin H and a U4/U6
snRNP-60K peptide.
J. Mol. Biol., 331: 45-56
DOI / PMID
Margittai, M., Fasshauer, D., Jahn, R., and Langen R.
Habc Domain and SNARE Core Complex Are Connected by a Flexible Linker.
Biochemistry, 42: 4009-14
DOI / PMID
2002
Fasshauer, D., Antonin, W., Subramaniam, V., and Jahn, R.
SNARE assembly and disassembly exhibit a pronounced hysteresis.
Nat. Struct. Biol. 9: 144-151
DOI / PMID / Commented in DOI / PMID
Antonin, W., Fasshauer, D., Becker, S., Jahn, R., and Schneider, T.R.
Crystal structure of the endosomal SNARE complex reveals common structural principles of
all SNAREs.
Nat. Struct. Biol. 9: 107-111
DOI / PMID
Pabst, S., Margittai, M., Vainius, D., Langen, R., Jahn, R., and Fasshauer, D
Rapid and selective binding to the synaptic SNARE complex suggests a modulatory role of
complexins in neuroexocytosis.
J. Biol. Chem. 277: 7838-48
DOI / PMID
2001
Margittai, M., Fasshauer, D., Pabst, S., Jahn, R., and Langen, R.
Homo- and heterooligomeric SNARE complexes studied by site-directed spin labeling.
J. Biol. Chem. 276: 13169-77
DOI / PMID
2000
Antonin, W, Holroyd, C, Fasshauer, D, Pabst, S, Fischer Von Mollard, G, Jahn, R.
A SNARE complex mediating fusion of late endosomes defines conserved properties of SNARE
structure and function.
EMBO J. 19:6453-64
DOI / PMID
Pabst, S., Hazzard, J.W., Antonin, W., Südhof, T.C., Jahn, R., Rizo, J., Fasshauer, D
Selective interaction of complexin with the neuronal SNARE complex: determination of the
binding regions.
J. Biol. Chem. 275:19808-18
DOI / PMID
1999
Davis, A.F., Bai, J., Fasshauer, D., Wolowick, M.J., Lewis, J.L., and Chapman, E.R.
Kinetics of Synaptotagmin Respones to Ca2+ and Assembly with the Core SNARE Complex onto
Membranes.
Neuron 24: 363-76
DOI / PMID
Fasshauer, D., Antonin, W., Margittai, M., Pabst, S., and Jahn, R.
Mixed and non-cognate SNARE complexes.
J. Biol. Chem. 274: 15440-46
DOI / PMID
1998
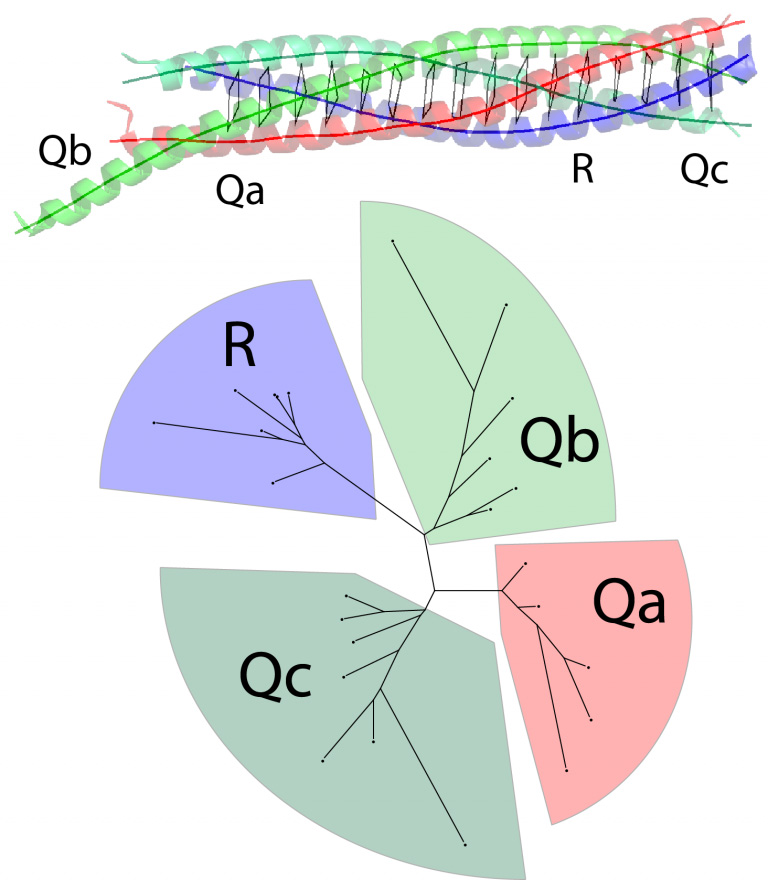
Fasshauer, D., Sutton, R.B., Brünger, A.T. and Jahn, R.
Conserved structural features of the synaptic fusion complex: SNARE proteins reclassified
as Q- and R-SNAREs.
Proc. Natl. Acad. Sci. USA 95: 15781-86
DOI / PMID
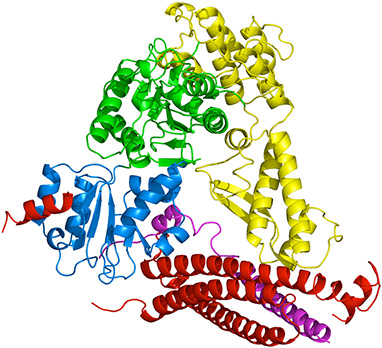
Sutton, R.B., Fasshauer, D., Jahn, R., and Brünger, A.T.
Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 Å resolution.
Nature 395: 347-353
DOI / PMID / Comment in DOI / PMID
Fasshauer, D., Eliason, W.K., Brünger, A.T., and Jahn, R.
Identification of a minimal core of the synaptic SNARE-complex sufficient for reversible
assembly and disassembly.
Biochemistry 37: 10345-55
DOI / PMID
1997
Fasshauer, D., Otto, H., Eliason, W. K., Jahn, R., and Brünger, A.T.
Structural changes are associated with SNARE complex formation.
J. Biol. Chem. 272, 28036-41
DOI / PMID
Fasshauer, D., Bruns, D., Shen, B., Jahn, R., and Brünger, A.T.
A structural change occurs upon binding of syntaxin to SNAP-25.
J. Biol. Chem. 272: 4582-90
DOI / PMID
1994
Nickel W, Kipper N, Barthel A, Kahn RA, Fasshauer D, Söling HD
ARF and VAPP14: two proteins involved in the delivery of heparan sulfate proteoglycan
from the trans-Golgi network to the plasma membrane.
Ann N Y Acad Sci. 733:344-56
DOI / PMID
Nickel W., Huber L.A., Kahn R.A., Kipper N., Barthel A., Fasshauer D., and Söling H.D.
ADP ribosylation factor and a 14-kD polypeptide are associated with heparan
sulfate-carrying post-trans-Golgi network secretory vesicles in rat hepatocytes.
J. Cell Biol. 125: 721-32
DOI / PMID